Automation
PRODUCTS
-
Viral vector manufacturing: key to successful gene therapy
Viral vector manufacturing, critical to the burgeoning field of gene therapy, is revolutionizing medicine with its rapid growth and significant potential for treating genetic disorders. By adhering to stringent cleanroom requirements, including BSL-2 standards, Germfree enables the safe and efficient production of viral vectors like AAV and lentivirus. Our specialized cleanroom solutions support the continued advancement of this vital therapeutic field, shaping the future of healthcare worldwide.
-
This model expands research possibilities, with a fourth laser option, multiple configurations and unique filter sets that allows investigators to focus on the science, not the instrumentation. Four different series are offered in this model. All instruments in the platform utilize CytExpert for CytoFLEX Acquisition and Analysis Software.
-
Buffer management solutions that reduce costs and expand possibilities.
Buffers play a significant role in biopharmaceutical manufacturing. They represent the major component by number and overall volume used in biopharma downstream processing steps. Buffer preparation makes up a substantial portion of a facility’s footprint and overall cost, labor requirements, and equipment.
-
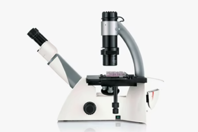
Just Right – Smart Choice
The DMi1 inverted microscope supports your specific work routine in your cell culture lab. Its operation is so intuitive, its handling so comfortable that you can fully concentrate on your work. Choose the functions you need, and, if necessary, you can easily add a variety of accessories that are important for your work.
-
Developing an optimized formulation tailored to your API nanoparticles is critical to unlocking their full potential.
WHITE PAPERS AND CASE STUDIES
-
Accelerating The Development Of Oncology Medicines
Oncology drug development has seen a significant shift in focus, as molecule chemistries and drug technologies have improved. Explore five areas for consideration and the potential emerging solutions.
-
Material Selection For Medical Device Manufacturability
Choosing the right material in the earliest stages of research & development can make all the difference in beating your competitors to market and avoid increased project costs, development delays and manufacturing setbacks.
-
A Lifecycle Management Strategy For Sterile Injectables
Gain insight into the benefits and challenges of transferring a therapeutic compound from one aseptic injectable format to another.
-
Neurovascular Device Case Study
Secant Group’s textile braiding and engineering expertise accelerates prototyping process for new neurovascular device in this case study.
-
Contextualizing Q Factor
An in-depth understanding of poles and zeros will improve your filter designs. Discover how open-source RF analysis tools can help you better understand how poles and zeros relate to Q factor.
-
Reducing Product Degradation With Pneumatic Conveying Systems
A leading multinational chain of coffee houses and roastery reserves encountered challenges when conveying their beans to a new roaster. Gain insight into the solution to their complex conveying needs.
-
The Keys To Bioabsorbable Process Development
Learn how custom tooling, material characterization, and expert guidance improved the production of bioabsorbable fixation sutures for an OEM, resulting in reduced IV loss and consistent shot-to-shot quality.
-
A New Challenge For Quality Experts – The Data Quality Concept
Quality experts are facing the challenge of rethinking their roles and redesigning the quality systems of their pharmaceutical companies to be based on the concepts of data quality.
-
Automated Data Capture To Train An AI Algorithm With eSource
Explore why an AI algorithm needs a steady and significant stream of data to become more accurate and prove its efficacy in a clinical setting.
-
Water For Life: ABB’s Measurement Technology Trailblazes Water Solution
Pioneering sustainable water supply with the world’s first dual mode desalination plant.
-
An Experienced CDMO Can Be A Differentiator In The Rapidly Growing Biologics Market
Biotech and biopharma developers are increasingly relying on outsourcing partners to meet both clinical and commercial research, development, and production needs. CDMOs that provide end-to-end services, including secure supply chains, can help biologics developers meet accelerated timelines and establish a real competitive advantage in today’s competitive market.
-
Version2 Gives Customers Direct Access To Information With Secure Documentation Solution
Version2 needed to consolidate all of their clients’ information into one place and automate everyday tasks. Keep reading to learn about how IT Glue helped Version2 create new standards and processes in their business with relationship mapping, integrations, flexible workflows, secure password access, and more.
NEWS
-
Copepods – Tiny Creatures That Can Help Reduce The Need For Soya Imports1/15/2024
In the sea, fish feed on species lower in the food chain. Can these same species form the basis of a new feed industry supplying the fish farming sector?
-
Hexagon Empowers Machine Shop Operational Excellence With Innovative New Software Suite9/20/2023
Hexagon’s Manufacturing Intelligence division announced today the release of HxGN Production Machining, a new suite of software developed to empower machine shops to achieve operational excellence in the manufacturing of discrete parts, tools, and components with machine tools at any scale, from one-off prototypes to volume production, and across industries ranging from medical to aerospace and beyond.
-
With Automated Treatment, Affordable Water From Nontraditional Sources Can Flow To Underserved Communities9/7/2023
Researchers at the Department of Energy’s Oak Ridge National Laboratory are developing advanced automation techniques for desalination and water treatment plants, enabling them to save while providing affordable drinking water to small, parched communities without high-quality water supplies.
-
National Resilience And Taiwan Bio-Manufacturing Corporation Announce Groundbreaking Partnership In Biomanufacturing Innovation2/5/2024
National Resilience, Inc. (Resilience), a technology-focused biomanufacturing company dedicated to broadening access to complex medicines, and Taiwan Bio-Manufacturing Corporation (TBMC), proudly announce a groundbreaking joint venture which aims to transform the biomanufacturing industry.
-
TRUMPF Lasers Drive Medical Technology2/8/2023
TRUMPF is an important partner for medical technology manufacturers: "Innovative medical devices cannot be manufactured without laser technology.