Networked Scanners
PRODUCTS
-
For successful regulatory outcomes, we provide quality medical writing services for clinical trial documents, safety updates, and marketing applications.
-
Germfree is internationally recognized as a leading manufacturer of Class II Biological Safety Cabinets. Our Class II Gloveboxes provide containment for diverse research applications in advanced biopharma laboratories. Our Class II BSCs are engineered to exceedingly rigorous standards for work that requires the protection of operator or product. We specialize in custom integration of all laboratory components.
-
Ensuring your computer software system validation efforts associated with manufacturing data workflow and materials management operations is our priority.
-
When selecting a 3D optical metrology solution, users should prioritize speed, measurement precision, magnification range, and the ability to measure surfaces from super-smooth to extremely rough.
-
Explore how an advanced Cell Line Development (CLD) platform for new biologics can enable CHO-based cell line development; ensure premium, stable, and high-yield results; overcome common expression bottlenecks; and provide robust verification, genomic characterization, and monoclonality assessment services.
WHITE PAPERS AND CASE STUDIES
-
Choosing The Right Fit For Your Business: Anatomy Of A Cash Drawer
The cash drawer has been a key component of the retail business for as long as people have exchanged money for goods. If you accept currency, you need the best cash drawer, but choosing the right drawer and till solution requires planning, consideration, and a little research.
-
Fujirebio Increases Study Agility By Streamlining Clinical Operations And Data Management Process
Fujirebio’s fast-growing U.S. Clinical R&D team was preparing for shorter studies with higher complexity. Traditional methods using paper and spreadsheet trackers were no longer adequate to meet quality goals. Learn how the group increased its agility and readiness with LifeSphere Clinical EDC and CTMS tools.
-
How Actigraphy Is Clearing Operational Barriers To Decentralized Trials
In this white paper, explore why sponsors are turning to proven systems for collecting real-world data to support their DCTs.
-
Partnering On An Innovative Hydrogel-Based Treatment For Glioblastoma
Treatments for glioblastoma multiforme (GBM) are in high demand. Learn more about the new possibilities for increased dosing and diffusion capabilities offered by nanoformed APIs.
-
Material Selection For Medical Device Manufacturability
Choosing the right material in the earliest stages of research & development can make all the difference in beating your competitors to market and avoid increased project costs, development delays and manufacturing setbacks.
-
Antibody Drug Conjugates: Global Trial Landscape - Focus On Asia Pacific
Gain a better understanding of the global clinical trial landscape of antibody drug conjugates (ADCs) and their trends in the Asia Pacific.
-
Centrifuges Transforming Biopharmaceutical Development And Manufacturing
Single-use centrifuges have the potential to become the new bioprocessing standard, helping manufacturers meet the need for innovative production methods when facing emerging modalities and next-generation biotherapeutics at scale.
-
The Role Of The Technical Transfer Executive Sponsor
Explore the importance of understanding a CDMO’s flexibility and culture, models and scale of equipment, technical capabilities, regulatory support, quality system, and executive sponsorship model.
-
ICH And USP <1220>: Implementing A Quality By Design Analytical Framework
The ICH and the USP are currently finalizing draft guidelines that describe a new paradigm for analytical development based on the QbD approach used for development.
-
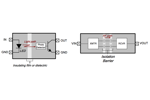
Isolator Vs. Optocoupler Technology
While optocouplers have been the dominant signal isolation solution for a long time, a new breed of higher-performance CMOS isolator solutions will ultimately out-mode the venerable optocoupler.
-
Oncolytic Virus Therapies – Global Clinical Trial Landscape - Focus On Asia Pacific
Explore the global landscape of oncolytic virus clinical trials, with a particular focus on trends in the Asia Pacific region.
-
Streamlining Study Start-Up For Accelerated Drug Development
Discover how we accelerated a Phase 1 study’s start-up time by 70%! This case study shares insights on streamlining communication, strategic planning, and more.
NEWS
-
Qorvo Delivers Superior Performance For 1.8 GHz DOCSIS 4.0 Cable Applications5/17/2023
Qorvo, a leading global provider of connectivity and power solutions, today announced the availability of its newest addition to the industry's most complete 1.8 GHz DOCSIS 4.0 product portfolio.
-
Opto Diode Introduces High-Performance Quad Electron Detector5/15/2023
Opto Diode Corporation introduces a high-performance quad electron detector, the AXUVPS7, with a total circular active area of 146 mm² (active area per element is 36.5 mm²).
-
Aclarity Recognized By Frost & Sullivan For PFAS Company Of The Year Award12/11/2023
Aclarity has been recognized by Frost & Sullivan as the 2023 Company of the Year, and was identified as best-in-class in the North American per- and polyfluoroalkyl (PFAS) substances treatment industry.
-
Xiao-I Launches Revolutionary Intelligent Document Processing Solution, Catalyzing The Future Of Enterprise Document Management6/19/2023
Xiao-I Corporation (Nasdaq: AIXI) (“Xiao-I” or the “Company”), a leading cognitive artificial intelligence (“AI”) enterprise in China, today announced the launch of its state-of-the-art Intelligent Document Processing (“IDP”) product.
-
Aratek Introduces The Marshall 8 Plus Biometric Tablet To Serve The Building Of Digital ID Ecosystems5/23/2023
Aratek recently launched the Marshall 8 Plus biometric enrolment tablet targeted squarely at helping governments and institutions build digital ID ecosystems for socio-economic development programs.