OCR
PRODUCTS
-
SearchLight™ is a free, intuitive, online plotting tool helps you overcome the obstacles many fluorescence instrument designers and users face with spectral management.
-
PHCbi brand's 3.0 cu.ft. (85 L) undercounter refrigerator is designed to be incorporated into ADA compliant workspaces and fits under most tables, cabinets, and benches. Its convenient glass door provides visibility into the interior of the unit to avoid unnecessary door openings. Temperature performance for your critical storage items in your tight spaces. Units can be stacked with an adapter to maximize valuable laboratory floor space. This space-saving lab refrigerator includes an automatic defrost function and is ENERGY STAR® certified.
-
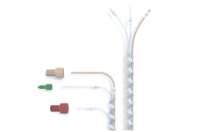
Medical tubing serves as the circulatory system for many devices, facilitating fluid transfer, drainage, and even electrical signal routing, but not all tubing is created equal. The materials, dimensions, tolerances, and chemical resistance must be meticulously designed to ensure patient safety and device performance.
-
Precision XY Saw Cutting offers high-volume, small-part fabrication with tight tolerances, ideal for optical components requiring precise dimensions and low defects.
-
For more than 20 years, Eurofins PSS Insourcing Solutions® has partnered with biotech and pharmaceutical companies to successfully validate and open new or remodeled facilities, meeting regulatory requirements. With a proven track record across numerous facility projects, our flexible, client-focused model ensures seamless integration with internal teams and third-party vendors, delivering high satisfaction and long-term partnerships.
WHITE PAPERS AND CASE STUDIES
-
Yorkshire Water Reduces Leaks By 57%, Eliminating 30% Of Annual Distribution Main Repairs
Yorkshire Water Services (YWS) is a leading UK water utility that serves nearly 5.5 million people and has a well-deserved reputation as a progressive and proactive utility. Their belief that innovation is one of the key driving forces that allows utilities to deliver better services to their customers while keeping costs down has led them to always seek out new ways to improve their operations both now and in the future.
-
Rapid Development Of A Liquid-Filled Capsule Formulation Of CBD
Discover how we developed and tested multiple oral formulations in just three months, identifying a top candidate that outperformed existing standards in key performance metrics.
-
Severn Trent Exceeds Leak Reduction Targets With Innovative Itron Water Solutions
Severn Trent’s smart metering program, powered by Itron’s technology, is transforming water conservation—reducing leaks by 6 million liters, cutting costs, and empowering customers to use water more wisely.
-
Inspecting On The Edge — Understanding Punch Tip Wear
Why is a horizontal optical comparator highly recommended for tablet manufacturers as a measuring tool for punch tip inspection, and what strategies can be used to detect, reduce, and prevent tip wear?
-
Advanced SWIR Sensor Development At SUI
Explore advancements in SWIR sensor technologies, enhancing time-of-flight imaging, avalanche photodetectors, and quenching circuits for high-performance applications in precision targeting, 3D LADAR, and photon detection.
-
EHR-To-EDC Success In A Complex, Adaptive Platform Trial
I-SPY 2, one of the longest-running adaptive platform trials in oncology, is revolutionizing breast cancer research through a dynamic, data-driven approach to evaluating novel therapies.
-
Electronic Batch Reporting
Learn how a CDMO automatically created a comprehensive batch report model to drive their reporting and speed root cause identification of quality failures with an Industrial DataOps software solution.
-
Optimizing Upstream Process Development To Increase mAb Titers
Uncover how we were able to help a client develop an efficient upstream process for a novel monoclonal antibody (mAb), resulting in a more than 100% increase in titers.
-
The City Of Fayetteville's Flood Resiliency In The Face Of Climate Change: Mapping 15 Watersheds
The city of Fayetteville, North Carolina has always had flooding issues, but it’s been getting worse as weather patterns have been changing. They were hit four years in a row by storms Matthew (2016), Irma (2017), Florence (2018), and Dorian (2019).
-
Re-Engineering A Complex Process For FDA Compliance
Discover the strategies that helped a company navigate a complex and shifting regulatory landscape to rapidly re-engineer a COVID-19 vaccine manufacturing process for FDA compliance.
-
How Optics And Flow Cytometry Are Improving Pancreatic Cancer Treatment
The University of Texas researchers are developing an optical system analyzing cancer cell properties to guide treatments, improving patient outcomes and targeting aggressive cancers like pancreatic.
-
Can Your Biopharma Team Keep Unplanned Downtime At Bay?
Learn how teams reduce downtime, strengthen compliance, and respond quickly to equipment issues through proactive maintenance, smart spare parts planning, and digital troubleshooting tools.
NEWS
-
Novotech Named Best Contract Research Organization In Australia At Asia-Pacific Biopharma Excellence Awards 20254/3/2025
Novotech, a globally recognized full-service clinical research organization (CRO) and scientific advisory company that provides biotech and small- to mid-sized pharma companies an accelerated path to market has been recognized as the Best Contract Research Organization in Australia at the Asia-Pacific Biopharma Excellence Awards (ABEA) 2025.
-
USTC Realizes Human Near-Infrared Color Vision Via Contact Lens Technology5/22/2025
A research team led by Prof. XUE Tian and Prof. MA Yuqian from the University of Science and Technology of China (USTC), in collaboration with multiple research groups, has successfully enabled human near-infrared (NIR) spatiotemporal color vision through upconversion contact lenses (UCLs).
-
Seeing The Unseen: Scientists Demonstrate Dual-Mode Color Generation From Invisible Light2/6/2026
Ultraviolet and near-infrared light are widely used in modern technologies but are invisible to the human eye.
-
A Breakthrough In Laser Cooling: Trapping Of A Stable Molecule With Deep Ultraviolet Light11/10/2025
Researchers from the Department of Molecular Physics at the Fritz Haber Institute have demonstrated the first magneto-optical trap of a stable ‘closed-shell’ molecule: aluminum monofluoride (AlF).
-
Schneider Optics Announces The Appointment Of Stuart Singer As A SPIE Fellow1/12/2026
Optics veteran Stuart Singer brings 45 years of leadership spanning lens design, manufacturing, vision systems, and cinema technology, with award-winning innovations and SPIE Fellow recognition.