Web Content Management
PRODUCTS
-
Conduct secure video based visits, manage study schedules, and keep participants connected through a single unified platform.
-
By removing barriers, and bringing trials to patients where they live, we can expand the geographical reach of clinical research across the globe allowing more patients to participate. Our patient-centric delivery means we can create convenience and comfort working the schedule of the trial around the schedule of the patient and their family, not the other way around. This approach provides important access to medical care, enhances the inclusivity of clinical trials and advances drug development for all.
-
The ORCA solution ensures consistent and precise chemical dosing with enhanced service and system automation to handle changing levels of hydrogen sulfide in real-time. This provides superior odor control performance and protects critical water infrastructure.
-
Code and approve clinical trial data from any device and keep your safety and efficacy data clean, consistent, and ready for analysis.
-
Design, build, simulate, and analyze clinical studies in one connected system. Embedded intelligence supports the full lifecycle, from protocol development to closeout.
WHITE PAPERS AND CASE STUDIES
-
Paradise Regained: How Digital Twins Saved A Bahamian Resort's Water Supply
Island utilities face unique resource constraints and infrastructure risks. Implementing digital twins provides the real-time visibility needed to reduce water loss, simulate emergency scenarios, and optimize system performance to ensure long-term operational resilience.
-
Digital Biomanufacturing, Now: From Digital Dreamer To Digital Doer
Discover how digital biomanufacturing is revolutionizing drug production by exploring strategies for integrating digital technologies to enhance efficiency and innovation in bioprocessing workflows.
-
How ABB's Flow And Digital Technologies Help Bawat With Smarter Ballast Water Compliance
The International Maritime Organization (IMO) sets strict global standards for ballast water management to prevent the spread of harmful aquatic organisms. For companies like Bawat, a Danish innovator in ballast water management, being able to verify ballast water flow measurements quickly, accurately, and from anywhere in the world is essential.
-
Expert Consulting And MRCT Strategy Rescues Oncology Program
Uncover how this strategic approach yielded significant outcomes, including uncovering missing data through IB review, streamlining dosing timelines, and achieving successful recruitment milestones.
-
Simplify CAPA In 7 Steps
Discover how to streamline corrective action/preventive action (CAPA) management in regulatory environments in 7 steps.
-
Revolutionizing Wastewater Management In Four Cities Through Water Mixing
Historically, lift station operators have turned to chemical treatments as the primary means of managing grease buildup. However, these methods carry inherent drawbacks.
-
Mobilizing Compliance: USP 797 And 800 Pharmacy Trailer Fleet For VA Hospitals
VA hospitals across the US are undergoing renovations to bring their facilities into compliance. The challenge was to design and manufacture a fleet of self-sufficient mobile compounding pharmacies.
-
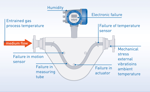
OPTICHECK Technology Built-In – Diagnostics, Verification, And Functional Safety With OPTIMASS Coriolis Mass Flow Meters
State-of-the-art measuring devices such as OPTIMASS Coriolis mass flow meters combine robust measuring principles and powerful electronics to produce a wide range of readings and device-specific data. OPTICHECK technology built-in leverages the features of the devices to translate their available comprehensive measuring system data into sophisticated diagnostics information for plant personnel.
-
Office Of Naval Research Contract Leads To New Chlorine Sensor Technology
Developed from U.S. Office of Naval Research requirements, this multiparameter chlorine sensor offers extended maintenance-free operation, flow-independence, and automatic compensation for reliable, in-pipe monitoring.
-
Rescuing Data Integrity: A Swift Transition And Quality Transformation
Uncover how this collaboration supported timely progress toward key study milestones, enabling smoother execution and more dependable output across data management and statistical activities.
-
Machine Learning In Intelligent Power Management Systems
Machine learning for MCU implementation (Tiny ML) is transforming battery management and motor control by utilizing machine learning algorithms to analyze sensor data, optimizing performance and assessing system health.
-
Enhancing Data Capabilities For Greenville Water Utility
The Greenville Water Utility in Indiana is leveraging Qatium to enhance its water management and leak detection capabilities, significantly improving service efficiency.
NEWS
-
Daxor Expands Blood Volume Analysis Technology Across Midwest, Wisconsin, And New Jersey Healthcare Networks3/28/2025
Daxor Corporation (Nasdaq: DXR), Daxor, the global leader in blood volume measurement technology, today announces a significant expansion into three regional healthcare facilities across the U.S. These facilities will use Daxor's Tennessee-based ezBVA Lab, a CLIA-certified facility equipped with state-of-the-art technology.
-
Athelas Strengthens Market Leadership In Strategic Partnership With Resilient Healthcare3/7/2025
Athelas, powered by Commure, announced a strategic partnership with Resilient Healthcare Corp, a trailblazer in health-at-home solutions, to launch a groundbreaking pilot program aimed at revolutionizing outpatient care and hospital billing.
-
Reusable Nanofiber Membrane Filters Water Sustainably10/23/2025
The antimicrobial triclosan is widely used in personal hygiene products, textiles and plastics, but when it enters the environment via wastewater, it poses a significant threat to aquatic organisms.
-
Eli Lilly's Foundayo™ Approved By FDA, Introducing A New Era Of Needle-Free GLP-1 Therapy4/1/2026
Foundayo, Lilly's second FDA-approved obesity medicine, will be available via LillyDirect® with free home delivery, starting at $25 per month with commercial coverage and $149 for self-pay
-
King Faisal Specialist Hospital And Research Centre Performs The World's First Robotic-Assisted Artificial Heart Pump Implantation1/16/2025
King Faisal Specialist Hospital and Research Centre (KFSHRC) in Riyadh has successfully performed the world’s first robotic-assisted implantation of an artificial heart pump (HeartMate 3) developed by Abbott, a groundbreaking procedure that marks a significant advancement in medical technology and patient care.