Business Process Management
PRODUCTS
-
Biotech companies progressing through clinical trials, planning an IPO, and undergoing rapid growth have a unique set of financial, contract, compliance, and reporting needs. We have designed an ERP solution, built on NetSuite, to address all of these needs. With over 50 implementations in the last two years alone, SuiteSuccess for Life Sciences is quickly becoming the defacto software standard for all successful biotechnology companies.
-
Scientific knowledge to strategize viable and efficient drug and device development pathways to achieve successful outcomes.
-
Recipharm Advanced Bio offers industry-leading biologics development services to support global clients in all clinical phases and global regulatory arenas. Our comprehensive development services include both process and formulation development.
-
NOVA™ Software Upgrade Kit adds Enhanced Microfeed with programmable rates down to 5 µm/min, plus pop-up prompts and improved folder/program management.
-
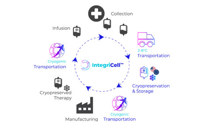
IntegriCell™ cryopreservation services from Cryoport Systems optimize leukapheresis-derived cell therapies by enhancing the safety, quality, and viability of manufacture-ready cryopreserved leukopaks. Our state-of-the-art cryopreservation process, integrated with our end-to-end global temperature-controlled supply chain platform, provides unmatched reliability, efficiency, and scope. Our standardized protocols meet the highest compliance standards, ensuring consistent quality and addressing product stability issues associated with fresh donor-derived cellular material for an optimized approach that enhances manufacturing efficiency and streamlines operations.
WHITE PAPERS AND CASE STUDIES
-
CDMO Uses Robotic Gloveless Isolator For Advanced Therapeutics
CDMOs must innovate production architectures to meet the demands of advanced therapeutics to ensure a reliable supply and compliance. Discover how these approaches advance modern medicine and improve patient outcomes.
-
Life Sciences Corporation Implements eLogbooks Across 15 Sites
New regulations challenged how surgical kits were tracked and processed across 15 U.S. sites. Discover how one team balanced compliance and efficiency to keep life-saving procedures on schedule.
-
CHO Cell Lines For Recombinant Protein Production
Discover how new strategies in CHO cell line development are driving innovation and transforming the future of biologics manufacturing, from enhanced productivity to improved therapeutic quality.
-
Preparing The Next Generation Of Clinical Research Professionals
Explore the live EDC platform that enabled Durham Tech to create a model for future-forward, hands-on education in clinical research.
-
Buffers And Process Liquids For Biopharmaceutical Production: Considerations For Scale-Up And Outsourcing
For small and midsize biopharmaceutical organizations, outsourcing buffer and process liquid preparation, quality testing, and storage could significantly streamline operations and reduce regulatory risk.
-
CHA Consulting Is Helping Florida Utilities Meet Ambitious Reclaimed Water Effluent Regulations
Discover how CHA Consulting has been working to help utilities across the sunshine state find the right balance and stay ahead of statewide deadlines for compliance.
-
End-To-End Workflow Integration For Antibody Development
Modern antibody discovery generates massive, fragmented datasets that slow collaboration and decision‑making. Examine how unified digital workflows streamline R&D and improve data quality.
-
GSK And Medable Discuss The Next Phase Of DCTs
GSK and Medable share the value of embedding DCTs into strategic goals to enhance trial accessibility, diversity, and efficiency.
-
Enhancing Data Capabilities For Greenville Water Utility
The Greenville Water Utility in Indiana is leveraging Qatium to enhance its water management and leak detection capabilities, significantly improving service efficiency.
-
Partner Case Study: Accelerating Gene Editing Manufacturing
Beam Therapeutics partnered with ElevateBio to manufacture BEAM-101, a novel base editing therapy for sickle cell disease, advancing rapid, high-quality production towards clinical trials.
-
How Sharp Packaging's Clinical Packaging Process Is 30% Faster
Discover how digitized workflows and real-time verification are helping streamline clinical trial packaging, reduce complexity, and improve quality in a high-stakes, time-sensitive environment.
-
A Risk-Based Approach To Plasmid DNA And mRNA Process Development
Balancing robust analytics and clinical readiness is key for early-phase pDNA and mRNA therapeutics amid structural complexity and regulatory challenges.
NEWS
-
Silvaco Expands Product Offering With Acquisition Of Cadence's Process Proximity Compensation Product Line3/4/2025
Silvaco Group, Inc. (Nasdaq: SVCO) (“Silvaco” or the “Company”), a provider of TCAD, EDA software and SIP solutions that enable semiconductor design and digital twin modeling through AI software and innovation, today announced the strategic acquisition of the Process Proximity Compensation (“PPC”) product line of Cadence (Nasdaq: CDNS).
-
FieldPulse Announces Integration With The Granite Group, Enhancing Efficiency For Contractors And Trade Professionals3/18/2025
FieldPulse has launched a new integration with The Granite Group.
-
Broadcom Launches VeloSky To Deliver Network Convergence, Transform Connectivity3/4/2025
Mobile World Congress 2025—Broadcom Inc. (NASDAQ: AVGO) today introduced VeloSky, a converged networking solution that enables Communications Service Providers (CSPs) to offer integrated fiber, cellular, and satellite connectivity through a single appliance.
-
SAP Unveils AI-Powered Innovations For Network-Centric Supply Chain Management5/22/2025
The innovations we unveiled represent a fundamental shift in how businesses can approach their operations in an increasingly complex global landscape.
-
Belden Unveils World's First 5G Industrial Switch Powered By Qualcomm At Hannover Messe4/13/2026
Belden Inc., a leading global provider of complete connection solutions, is showcasing a groundbreaking demonstration of the world's first 5G industrial switch specifically designed for use on the factory floor.