Compliance
PRODUCTS
-
Precise, Complete, and Ultimate Detection.
A premium dual energy x-ray inspection system offering outstanding detection of hard-to-find, low density contaminants, in a variety of applications. Available in standard and custom configurations
Detect Hard-to-Find Contaminants
Capable of detecting hard-to-find contaminants in a variety of applications. Offering clarity and precision in complex areas of product inspection.
Enhance Product Quality
Achieves outstanding product quality using advanced levels of foreign body detection of low-density contaminants in overlapping products.
Increase Productivity
Clever design combined with an intuitive user interface to create user-friendly systems that increase uptime and boost productivity.
-
The AMP20072 is a solid-state high power amplifier designed for a wide range of applications including EMI/RFI testing, laboratory work, continuous wave (CW) and pulse operations, and general communication use.
-
For more than 20 years, Eurofins PSS Insourcing Solutions® has partnered with biotech and pharmaceutical companies to successfully validate and open new or remodeled facilities, meeting regulatory requirements. With a proven track record across numerous facility projects, our flexible, client-focused model ensures seamless integration with internal teams and third-party vendors, delivering high satisfaction and long-term partnerships.
-
Meet FDA guidelines for GMP in tablet and capsule production with a secure, failsafe system. Qualification reports are available.
-
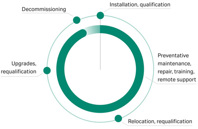
Whether your Cytiva instrument is brand new or well-seasoned you’ll have trained help at the ready. Explore the services we provide for each stage of your equipment’s life.
WHITE PAPERS AND CASE STUDIES
-
Preparing For Post-Market Clinical Follow-Up Under EU MDR
Gain a better understanding of the primary challenges faced by the life sciences industry's commercial model in response to the updated EU MDR guidelines.
-
GFI Case Study: Elgin Community College Uses GFI MailArchiver To Comply With E-Discovery Laws Elgin Community College is an educational institution in Elgin, Illinois offering first-class education from certificate to degree level. Changes to compliance and e-discovery regulations meant that the college required a system that allowed it to be in a position to provide electronic data should it be required as evidence in a civil or criminal legal case. To this end, the College installed GFI MailArchiver, an email management and archiving software.
-
InnovaGel Gains Quick Return On Investment
InnovaGel, a manufacturer of vitamins and supplements in the nutraceutical and OTC market, was in need of solution for its soft gelatin encapsulation machine application.
-
Case Study: Workforce Management Solution Supports Smart Meter Rollout During the technology evaluation stage, CHED Services recognized an opportunity to derive further benefits from the launch of a proven enterprise workforce management system. By Ventyx
-
How To Survive An FDA Audit
Understanding the FDA audit process, preparing for it and being aware of regulations will help to restore a sense of control and help to ensure the best outcome.
-
Developing An Organizational Roadmap For Post Authorization Safety Studies
Delve into this case study to find out how a company found a partner to help them analyze the current state of their pharmacovigilance systems, identify opportunities for improvement, and implement industry best practices and standards.
-
Ensuring On-Time Release Of CAR-T Cell Treatment In Spain
Due to the COVID-19 pandemic, and the start of Brexit, a CAR-T cell treatment therapy shipment was offloaded multiple times by the airline, requiring an alternative transport solution.
-
Simple SOP Updates Every Research Site Needs To Succeed At Remote Monitoring
Today’s crisis climate is creating a dichotomy in clinical research. As a result, sites are closing, medical facilities are enforcing access restrictions, and there are constant delays or stoppage of non-essential studies. On the other hand, this health crisis is highlighting the critical need for clinical research due to ongoing COVID-19 trials. As a result, clinical researchers are asking questions and seeking workarounds to continue operations in response to site-specific adjustments. One central issue has surfaced — how do we continue to maintain compliance when working from home?
-
Distribution Of The First Commercial CAR-T Therapy From USA To Japan
Examine the first commercial live shipment of temperature-critical autologous final CAR-T drug product from a USA manufacturing site to a hospital in Japan.
-
Physicians Medical Group Cuts Processing Time With Datacap Taskmaster Physicians Medical Group of Santa Cruz County (PMG) cut its claims processing time by more than half and provided faster access to data, fewer compliance concerns, happier employees and more satisfied customers with an integrated solution from Datacap and Bowe Bell & Howell. Submitted by Datacap
-
Commercial Metals Company Centralizes AP Processes And Save 55,000 A Year With ImageSilo® When the Director of Shared Services at Commercial Metals Company (CMC) was asked to centralize the accounts payable processes of 40 metal recycling yards scattered across the country, he quickly realized that paper documents were the roots of his AP problems at the yards. By Digitech Systems, Inc.
-
Supporting the Pharmaceutical Industry with 21 CFR Part 11 Compliance Readiness The purpose of this document is to describe how PANalytical systems support system owners meeting the requirements of the 21 CFR Part 11 regulations issued by the United States’ FDA. Submitted by PANalytical
NEWS
-
Curia Invests $4M To Enhance Sterile API Manufacturing10/27/2025
Curia, a leading global research, development and manufacturing organization, today announced the completion of a $4M investment to upgrade its two API aseptic suites in Valladolid, Spain.
-
Zipline Announces UKG Partnership Enhancement6/17/2025
Zipline, the leading platform for frontline execution and communication, today announced a major enhancement to its UKG integration. With this release, frontline employees can now confirm key compliance-related details directly in Zipline during clock-in and clock-out, with the information syncing seamlessly to UKG’s existing attestation workflows.
-
MangoApps Adds AI-Powered Scheduling & Shift Swap To Its Extensive Workplace Operations App Marketplace7/15/2025
MangoApps today announced powerful new enhancements to its workplace operations platform, introducing AI-powered scheduling and intelligent shift swap capabilities.
-
Oncotelic Therapeutics Announces Strategic Partnership With TechForce Robotics To Commercialize PDAOAI-Enhanced GMP Robotics Platform4/2/2026
Oncotelic Therapeutics, Inc. (“Oncotelic” or the “Company”), a clinical-stage biotechnology company focused on oncology and AI-driven solutions, today announced that it has entered into a strategic partnership with TechForce Robotics, Inc. (“TechForce”) to advance the commercialization of its PDAOAI-enabled, GMP-compliant robotics platform.
-
Paylocity Advances One Unified HCM And Finance Platform With Launch Of Integrated Spend Management Solution7/22/2025
Paylocity, a leading provider of cloud-based HR, payroll and spend management solutions, today announced the launch of Paylocity for Finance, a strategic expansion of its modern workforce platform into the Office of the CFO.